
- This event has passed.
Professor Andrew Murkin Seminar
April 21, 2017 @ 1:00 pm - 2:00 pm

Dr. Andrew Murkin, from University at Buffalo, The State University of New York, will present a seminar titled, “Mechanism and inhibition of enzyme-catalyzed retro-aldol reactions in Mycobacterium tuberculosis.”
Abstract:
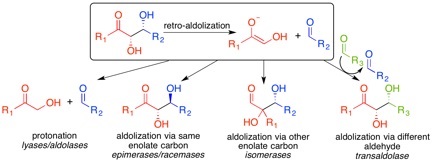
Retro-aldol reactions involve the cleavage of a compound containing an alcohol b to a carbonyl, yielding an aldehyde and another carbonyl-containing product (i.e., aldehyde, ketone, ester, or carboxylic acid). Enzymes that catalyze this process may release the two products, as in the case of aldolases and other carbon-carbon lyases, or may utilize the fragments as intermediates in transfer, epimerization, and isomerization reactions:
Isocitrate lyase (ICL) and 1-deoxy-D-xylulose-5-phosphate (DXP) reductoisomerase (DXR) are two drug targets from Mycobacterium tuberculosis that are believed to employ retro-aldol steps in their reactions. Mechanistic details have been revealed through the application of steady-state kinetics and kinetic isotope effects (KIEs). Additionally, 3-nitropropionate, a substrate analogue previously reported to be a potent reversible inhibitor of ICL, has surprisingly been found to act as a covalent inhibitor of the enzyme. Armed with this mechanistic information, we are designing inhibitors that will hopefully provide new opportunities for drug discovery against this evasive organism.
Host: Patrick Frantom